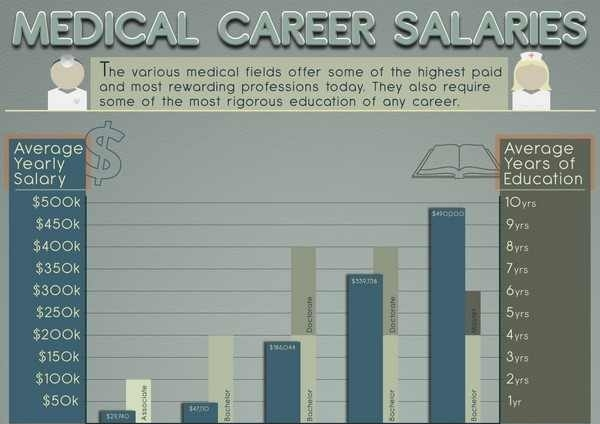
Under the supervision of the Director of Clinical Data Disclosure & Transparency, the position is responsible to: The Specialist – Clinical Data Disclosure & Transparency Medical Writer position will have primary responsibility for preparing trial disclosure documents in compliance with company policy and country laws and requirements. We are on the leading-edge of healthcare breakthroughs that help provide new, reliable, and compliant medical products, practices and solutions to the world. Our Regulatory Affairs team bring new medical advancements to the world by facilitating communications and procedures that allow swift, organized compliance partnering with external regulatory agencies.  
However, subject to applicable law, employees working in roles that the Company determines require routine collaboration with external stakeholders, such as employees in customer facing commercial or research based roles, will be required to be fully vaccinated as a condition of employment. New hires in office-based roles in the US & Puerto Rico will be required, subject to applicable law, to demonstrate that they have been fully vaccinated for COVID-19 or qualify for a medical or religious exemption to this vaccination requirement that can be accommodated without an undue burden to the operation. Specialist-Transparency/Disclosure Medical WriterĪt Merck in Dover, Delaware, United States
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
